
Titanium and its alloys tend to naturally grow a homogeneous oxidized layer, which shows low electrical conductivity, thermodynamic stability and lesser tendency to form ions. Even though its thickness does not exceed 10 nm, it gives extra protection against corrosion to the surface.
Anodizing is an electrochemical process used for surface treatment and finish. The process requires the element to be immersed in an electrolyte solution and acting as the anode. Then, applied electrical current splits water molecules into oxygen and hydrogen in a process called electrolysis. As the current passes, it forces oxygen atoms to the anode (titanium element) which build up the layer of titanium oxide. The layer grows gradually and its thickness depends on the applied voltage.
Anodizing is used for the following purposes:
- protection against corrosion, as well as wear and tear,
- increased abrasion protection,
- to create a colorized layer on the surface,
- to create an underlayer for dye or organic coating,
- to get a protective, thermal barrier.
Not all layers achieved through anodizing are the same – the process of color anodizing differs from the one used for wear protection purposes.
Color anodizing is commonly used in the medical industry, to help visually identify the parts. In jewelry manufacturing, anodizing is also used for aesthetic purposes. Aviation and military industries along with tool-making businesses relay on anodizing to improve mechanical and physical properties of the elements made out of titanium.

Norms related with titanium anodizing:
- AMS 2488 – describes the type of anodizing which is performed in an electrolyte with pH value higher than 13.
Anodizing in base environment creates thicker oxygen coating and can be considered as a “heavy duty” protection against wear and abrasion.
The norm identifies two types of anodizing – type 2 and type 3 (as type 1 is far less common).
Type 2 – wear and tear purposes,
Type 3 – also called “color anodizing”, widely used in the medical, aviation or military industries.
Type 2 anodizing does not create color variations as anodized surface is a distinctive grey color and it helps to prevent friction between sliding titanium surfaces. - AMS 2487 – this norm specifies the properties of oxidized layer as well as describes the requirements for electrochemical process for electrolytes with pH value not exceeding 12,4. These coatings are classified as abrasion resistant.
Anodized layers can be also classified based on electrolytes used during the process. Layers made in sulfuric acid or chromic acid solutions are porous. Phosphoric acid shows higher dissolving properties, which makes the anodized layer even more porous; such layer is used for adhesion of titanium with other metals. Weaker electrolytes, such as tartaric acid, ammonium tartrate, boric acid or citric acid would influence the oxidized layer far less. Such layers are non-porous and create a impenetrable barriers.

Color anodizing
Color coating is obtained through the phenomenon called optical interference, as the light reflects from both – the oxide layer as well as underlying titanium surface at different angles, making them interfere with each other (intensifying or cancelling the light wavelengths), giving the illusion of color – just like a prism. By increasing the voltage, the layer builds thicker, changing the refraction of light as thickness of the layer changes the color.
Galvanic cell for anodizing titanium
Before anodizing process, all mechanical treatment of the element’s surface must be concluded. The layer giving the color is very thin and could be easily removed by polishing or sanding. The finish of the surface has a significant influence on refraction of light, for example matt finish would give darker colors, however the color will not change due to angle of view as unpolished surface refracts light more. Polished, shiny surface would give much lighter and “live” colors, but the angle of viewing may influence the shade. Before anodizing process, make sure the surface is clean from dust, grease or other contaminants which may influence the process.
Attention: Tap water includes minerals, which may and will cause defects in anodized oxidized layer.
Make sure that the surface of cathode is equal or bigger than that of anode. In the best case scenario, cathode should surround anodized element. Insert plastic protective nets between cathode and the walls of the container.
Rainbow effect
To obtain the rainbow effect, immerse the element in electrolyte. By adjusting the voltage, you can influence the color of the element in a galvanic bath. Slowly emerge the piece from the solution and at the same time, increase the voltage.
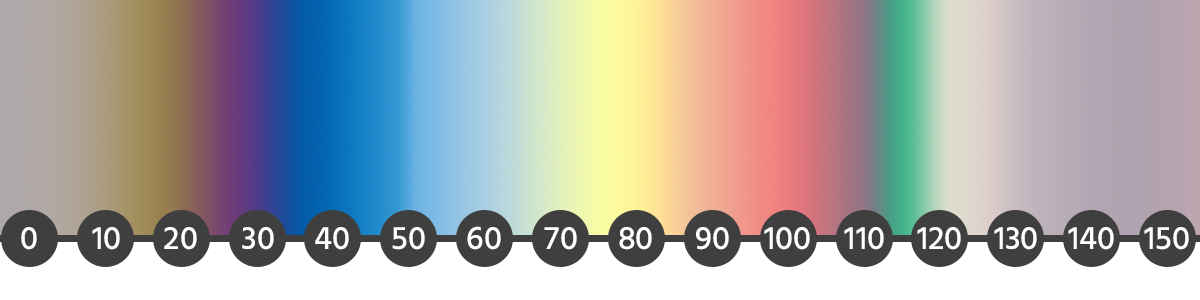
How to make distinct, multicolor patterns
To make a polychromatic patterns with distinct, uniform colors, you would need to use a tape during anodizing process. The tape prevents the electrolyte to react with the covered surface, while the exposed one undergoes the anodizing process. Start with the surfaces which require the highest voltage. When the desired color is obtained, remove the tape and immerse the element again, and this time apply lower voltage. Lower voltage has no influence on the color and surface already treated with higher voltage.

Other articles
Norms and standards of non-ferrous metal products
All non-ferrous metal products offered by WOLFTEN are manufactured according to specific standards. Clearly defined technical standards guarantee the homogeneity of the materials used, which directly affects the specific characteristics of each structure and their safety.
Sustainable environmental technologies and metallurgy
Can metallurgy be considered a sustainable environmental technology? This is the question that bothers not only environmental activists, but anybody concerned with the mining industry having a considerable impact on the natural environment.
Neural networks in metallurgy
Ability to develop, create and apply a specific material marks the border between business success or failure. Forecasting mechanical properties of a new alloy is an important aspect for both scientists and engineers as it allows saving time and money.



